The government says it’s confident an Indian-made AZ vaccine used in the UK won’t affect people’s ability to travel abroad.

image copyrightGetty Images
The government says it’s confident travel to EU countries by UK citizens – jabbed with an Indian-made AstraZeneca vaccine – won’t be affected, even though the Indian manufacturer has not been approved by the European Union.
In India, the AstraZeneca (AZ) jab is made by the Serum Institute – normally under a different brand-name, Covishield.
The Department for Health and Social Care says no Covishield vaccines have been administered in the UK.
Most of the UK’s supplies of the AZ vaccine come from plants in north Wales and Staffordshire.
But five million doses were shipped from India as part of the UK’s 100-million-dose AZ order.
The batches were produced by the Serum Institute of India (SII) – but the Department for Health says they were not called Covishield.
An amendment was made to the agreement between AstraZeneca and the MHRA (the UK regulator) listing three SII batch numbers and stating they were being “assessed and are treated as Covid-19 Vaccine AstraZeneca”.
The batch numbers are 4120Z001, 4120Z002 and 4120Z003.
If you received the AZ vaccine, it’s possible,
A quick way of checking which batch you have had is to look at the paper card you were given after vaccination.
The person who jabbed you should have written the batch numbers in boxes on the back.
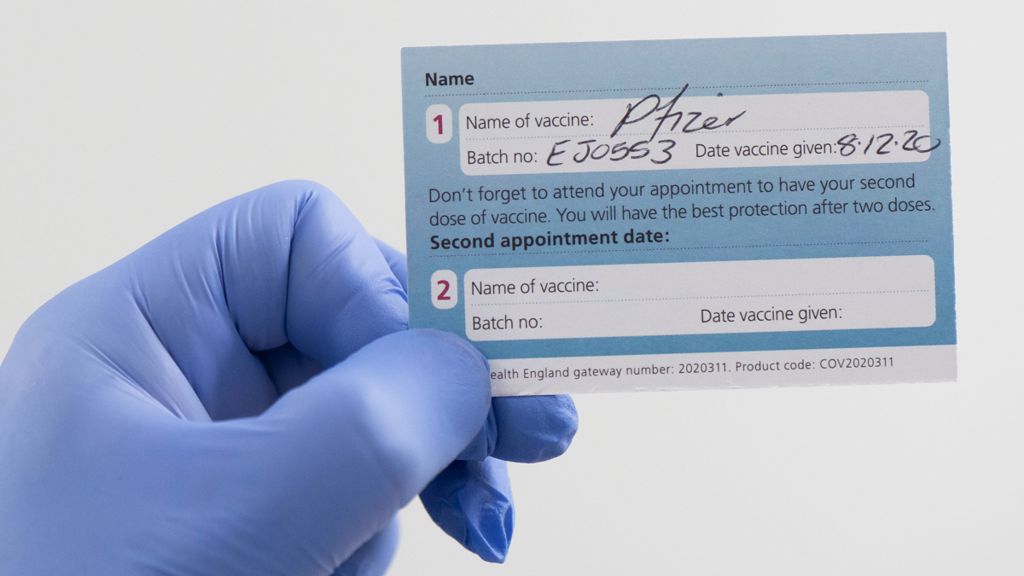
image copyrightGetty Images
You will also see batch numbers listed on NHS Covid Pass letters which show you are fully vaccinated, and which you will need when travelling abroad to some countries.
You can see a digital version of the letter on the NHS App.

NHS letters for international travel will also have the AZ jab listed as Vaxzevria.
That’s the AstraZeneca vaccine name recognised by the EU’s medicine regulator – the European Medicines Agency (EMA).
Other vaccines approved by both the EU and the UK are also sometimes known by different names:
- Oxford-AstraZeneca (AZ) (also known as Vaxzevria)
- Pfizer-BioNTech (also known as Comirnaty)
- Moderna (also known as Spikevax)
- Janssen (sometimes referred to as the Johnson & Johnson vaccine) is the same as in the UK
Your paperwork may show Indian-made batch numbers, but the Department for Health says: “All AstraZeneca vaccines given in the UK are the same product and appear on the NHS Covid Pass as Vaxzevria.”
“The European Medicines Agency has authorised this vaccine and we’re confident travel will not be affected.”
The NHS Covid Pass is not yet accepted by the EU as bloc – but some individual countries, such as Greece and Spain, are already accepting it.
The government hopes it will soon be accepted as a “vaccine passport” – letting people cross borders more easily, with less need for quarantine.
The EU says it’s working to ensure its own version – the EU Digital Covid Certificate – is compatible with similar products in non-EU countries.
If it’s satisfied the NHS pass complies with EU standards and systems, it can decide to accept it.
It can be accessed via the existing NHS app.
For people aged 16 and over in England, the digital version shows proof of:
- vaccination
- a negative PCR or lateral flow test in the past 48 hours
- recovery from Covid-19
A paper version confirming you have been double-vaccinated can be requested online or by calling 119.
Paper versions are available for people in Scotland, Wales and, from early to mid-July, Northern Ireland.
The European Union’s vaccine “passport” is being rolled out across all 27 member nations – plus Switzerland, Iceland, Norway and Liechtenstein.
The certificate can be issued if a citizen has:
- been vaccinated against Covid-19
- recently had a negative PCR test for the virus
- recently recovered from Covid-19
Anyone holding a certificate should, in principle, be exempted from testing or quarantine when crossing an international border.

image copyrightGetty Images
It has a quick-response (QR) code with a digital signature key unique to the place an individual’s vaccine information is stored – a hospital, test centre or health authority, for example.
The individual’s data remains on the certificate and is not stored or retained when it’s verified – at an airport, for example.
Some countries had already been using the certificate on a voluntary basis – but it was officially introduced on 1 July with a six-week phase-in period.
Only EU citizens, as well as non-EU nationals legally staying or living in the member states (with the right to travel to other member states), can download it or obtain a paper copy – at no cost.
It is not a travel document and travellers will still need to carry a passport or another form of identification.
If you are an EU national who was vaccinated in the UK, it is up to your home country whether you are eligible for the EU certificate.
The Indian product – widely used in poorer countries via the Covax vaccine programme – is identical to the one made in Europe.
But the EMA hasn’t needed to approve it because the EU isn’t receiving any doses from the site in India where it’s manufactured.
However, the World Health Organization has approved Covishield for emergency use, and the European Commission says individual member states may agree to let in travellers fully-vaccinated with Covishield.
Some countries already accept other vaccines – for example, Greece accepts China’s Sinovac, Russia’s Sputnik V and several others.
The Serum Institute is understood to be seeking emergency EU authorisation for Covishield.
AstraZeneca also says it’s working with the EMA on the “inclusion of Covishield as a recognised vaccine for immunisation passports” – although the EMA says there is currently no application for market authorisation.

